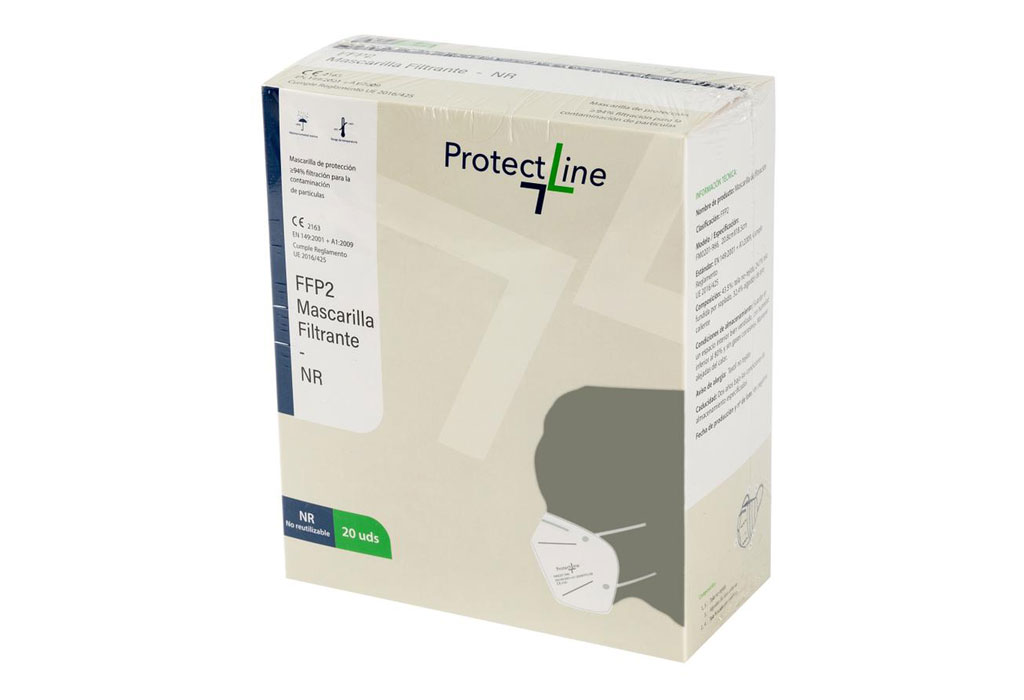
FFP2 (NR) masks
MODELS: FM0201-966R
2163
FFP2 masks are an Individual Protection Equipment whose purpose is to filter the inhaled air avoiding the entry of polluting particles into our body.
Our masks are CE marked and include the reference to the UNE EN-149 standard, which ensures compliance with a quality standard. They also incorporate the NR marking, which indicates that the product is Non-Reusable.
- Product Name: Filtering Mask
- Classification: FFP2
- Model / Specification: FM0201-966, 20.8cmX18.5cm
- Standard: EN 149: 2001 + A1: 2009, Complies with EU Regulation 2016/425
- Composition: 43.5%: non-woven fabric, 24.1% melt blown fabric, 32.4% hot air cotton
- Storage conditions: Store in a well-ventilated indoor space, with humidity below 80% and without gases corrosive. Keep away from heat
- Allergy Notice: Non-woven Textile
- Expiration: Two years under specified storage conditions
- NR (Non-Reusable) equipment should not be used in more than one work shift.
- Use under the manufacturer’s recommendations, following the labeling instructions.
- Non-medical use. Not sterile.
- These masks do not provide oxygen.
- Do not use in atmospheres with low oxygen concentration or poorly ventilated areas.
- The presence of special facial features, abundant hair or glasses can prevent a correct fit of the mask.
- Keep away from heat sources.
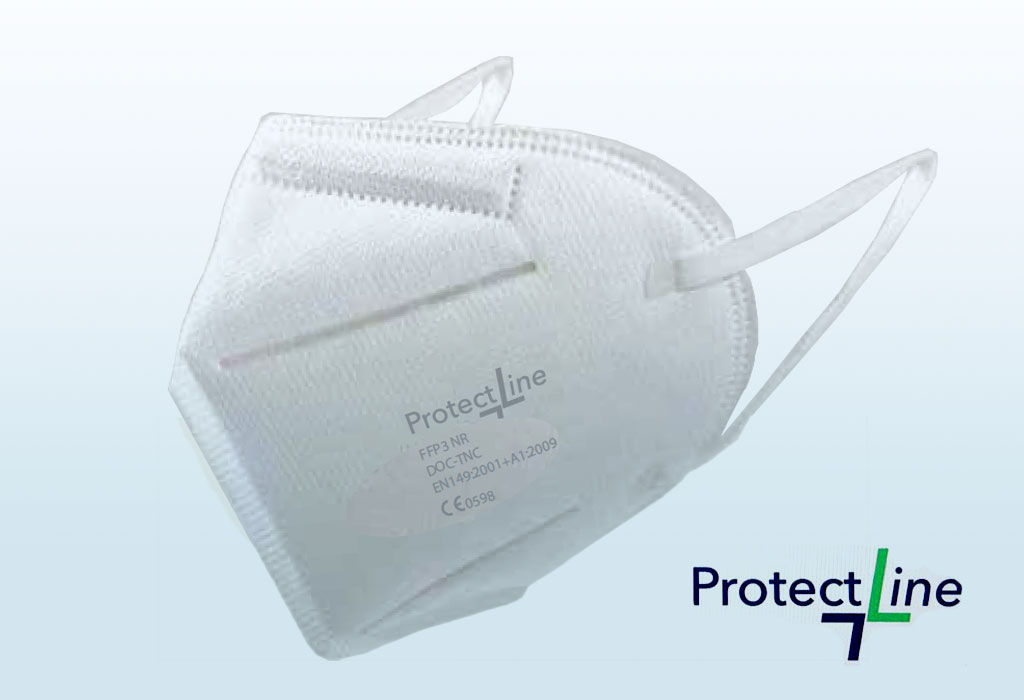
FFP3 (NR) masks
MODELS: DOC-TNC MODEL
0598
FFP3 masks are Individual Protection Equipment whose purpose is to filter the inhaled air avoiding the entry of polluting particles into our body.
Our masks are CE marked and include the reference to the UNE EN-149 standard, which ensures compliance with a quality standard. They also incorporate the NR marking, which indicates that the product is Non-Reusable.
- Product Name: Filtering Mask
- Classification: FFP3
- Model / Specification: DOC-TNC , 15cm x 10.5cm
- Standard: EN 149: 2001 + A1: 2009, Complies with EU Regulation 2016/425
- Composition: 55.9%: non-woven fabric, 29.4% melt blown fabric, 14.7% hot air cotton
- Storage conditions: Store in a well-ventilated indoor space, with humidity below 80% and without gases corrosive. Keep away from heat
- Allergy Notice: Non-woven Textile
- Expiration: Two years under specified storage conditions
- NR (Non-Reusable) equipment should not be used in more than one work shift.
- Use under the manufacturer’s recommendations, following the labeling instructions.
- Non-medical use. Not sterile.
- These masks do not provide oxygen.
- Do not use in atmospheres with low oxygen concentration or poorly ventilated areas.
- The presence of special facial features, abundant hair or glasses can prevent a correct fit of the mask.
- Keep away from heat sources.
Surgical Mask
MODEL FM0501-966
Surgical masks are Individual Protection Equipment whose purpose is to filter the exhaled air in order to protect those around you, avoiding the spread of viruses when sneezing, coughing or speaking.
Our surgical masks have the EC label which ensures that the product complies with associated the legislation; the reference to the UNE EN 14683 standard, which ensures compliance with a quality standard, and whether it is a Type I or a Type II.
- Product Name: Disposable Surgical Mask – Type IIR
- Classification: Surgical, I, Rule 1
- Model / Specification: FM0501-966, 17.5cm x 9.5cm
- Specification: Single use surgical masks. Requirements for materials, design, preparation, marking and use. Part 1: For use in adults
- Standard: UNE-EN 14683: 2019 AC
- Bacterial filtration efficiency :> 98%
- Composition: 61.5% non-woven fabric, 38.5% melt blown fabric
- Structure: mask body, nose clip and mask grip
- Allergy Notice: Non-woven Textile
- Expiration: 12 months under specified storage conditions
- Hypoallergenic, polypropylene and waterproof material including flexible nose clip.
- CE Test Report issued by TÜV SUD.
- Warning: This device is not a medical device within the meaning of Directive 93/42 or Regulation EU/2017/745, nor is it a personal protective equipment within the meaning of European Regulation EU/2016/425 ″.
- Conservation conditions: Store in a place with relatively low humidity, free of corrosive gases, well ventilated and avoiding high temperatures (> 40ºC).
- Mask for personal use.
- Maximum duration of use: For reasons of comfort and hygiene, it is recommended not to use the mask for more than 4 hours. It may be used several times, provided that it is always removed following the Non Reusable instructions.
- Not Reusable.
- Disposal of the mask: Masks should be disposed of in a container provided with a plastic bag (preferably with a lid and non-manual control). It is recommended to use a double bag to preserve the contents of the first bag in case of tearing of the outer bag.
Covid-19 test
TYPES OF TEST
PCR: detects the genetic material of SARS Cov-2 coronavirus, following the patterns already been used for other viruses. They take mucous samples and are analyzed in laboratories. Results take between three and six hours to be obtained.
Antigen test: The COVID-19 Rapid Antigen Test is a solid phase immunochromatographic test for the qualitative in vitro detection of new coronavirus 2019 antigens in human nasopharyngeal or oropharyngeal secretions. This test provides only a preliminary result of COVID-19 infection as a clinically assisted diagnosis. The test is applicable to the clinical system, medical institutions and the field of scientific research.
IgG / IgM antibody test: The COVID-19 IgG / IgM Rapid test Kit (Blood / Serum / Plasma) is a solid phase immunochromatographic assay for the rapid, qualitative and differentiated detection of IgG and IgM antibodies against the new coronavirus in blood, plasma or blood serum . This test only produces a preliminary result. Therefore, any sample that reacts to the COVID-19 IgG / IgM Rapid test Kit must be confirmed with an alternative method and with clinical findings.
Protective suits and gowns
Our protective suits and gowns are designed for healthcare personnel to protect themselves from drops of coughs, sneezes or other bodily fluids from infected patients and from contaminated surfaces that may infect them.
The protective suits comply with the UNE-EN 14126: 2004 standard, which contemplates specific tests of resistance to the penetration of microorganisms.
This type of clothing can offer different levels of tightness both in its material and in its design, partially covering the body such as gowns, aprons, sleeves, gaiters, etc., or the entire body. The designation includes the Type and the letter B (for Biological).
Our gowns comply with the 93/42 / CEE standard.
0123
Contact us
Do you have any questions about our services? You can call us or send us an email.